
The negative sign in Equation 7.3.3 indicates that the electron-nucleus pair is more tightly bound when they are near each other than when they are far apart. An absorption spectrum results when light from a continuous source passes through a cooler gas, consisting of a series of dark lines characteristic of the composition of the gas. The orbit with n = 1 is the lowest lying and most tightly bound. An emission spectrum can be produced by a gas at low pressure excited by heat or by collisions with electrons. This spectrum contains dark lines on bright. In his final years, he devoted himself to the peaceful application of atomic physics and to resolving political problems arising from the development of atomic weapons.Īs n decreases, the energy holding the electron and the nucleus together becomes increasingly negative, the radius of the orbit shrinks and more energy is needed to ionize the atom. Absorption spectrum : The spectrum produced by absorbed radiation is known as absorption spectrum. Stellar spectra are approximately a black body spectrum, but this mostly holds true for the most hot and luminous O and B type stars for cooler stars, the image already gets quite a bit more muddled, as they will show more and more absorption happening in the stellar photosphere, as well as other effects that ruin the nice, smooth black body sp. (b) The energy of the orbit becomes increasingly less negative with increasing n.ĭuring the Nazi occupation of Denmark in World War II, Bohr escaped to the United States, where he became associated with the Atomic Energy Project. The absorption spectrum is obtained when the substance absorbs the. The atom has been ionized.įigure 7.3.2 The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. 1) The emission spectrum is obtained due to emission of radiation from the substances. In this state the radius of the orbit is also infinite. In that level, the electron is unbound from the nucleus and the atom has been separated into a negatively charged (the electron) and a positively charged (the nucleus) ion. In this model n = ∞ corresponds to the level where the energy holding the electron and the nucleus together is zero. Where \( \Re \) is the Rydberg constant, h is Planck’s constant, c is the speed of light, and n is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus. The diagram shows the energy levels in a hydrogen atom.Īn orbiting electron has less energy than a free electron, so the energies are shown as negatives relative to the ground state.Īn electron with the minimum possible energy is in the ground state higher energy levels are called excited states.\] In such a case, apparently, an aureole containing atomic hydrogen is formed at the near end of the capillary and acts as an absorbing layer. The adsorption ( ads 1.0 ± 0.2 eV) and absorption ( abs 0.1 ± 0.2 eV) energies predicted by DFT depend strongly on the choice of functionals. The energies are measured from a zero equivalent to a single free electron. Line spectrum are unique for each element, and for each isotope of that element.Īn energy level diagram shows the amounts of energy that electrons have at each level in an atom. When current is passed through hydrogen gas, the hydrogen spectrum is given as below: The samples emission spectrum will be missing from the result due to the absorption of energy by electrons corresponding to the energy levels of the element. Emission or absorption processes in hydrogen give rise to series, which are sequences of lines corresponding to atomic transitions, each ending or beginning. Line emission and absorption spectra Studying the line spectra produced by hot gases and absorbed by cooler gases allows us to identify the elements in stars. 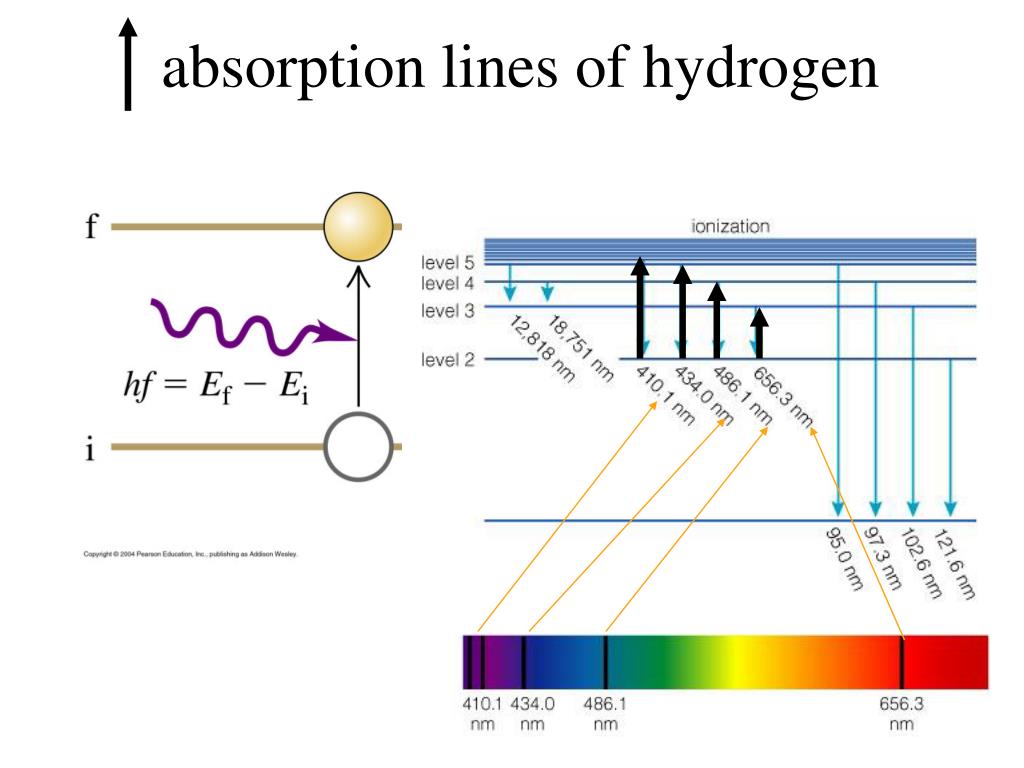
The energy is lost in the form of photons of certain wavelengths, which are different for each material.Ī Line Spectrum can be create by passing current through an ionised gas.Īn Absorption Spectrum is produced by shining white light through a sample of a gaseous element. Line Spectra are the wavelengths of light emitted from an element when it's electrons lose energy.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
